TRICHLOROMETHANESULFONYL CHLORIDE
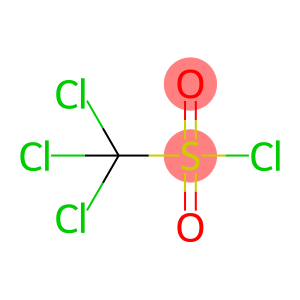
Trichloromethanesulfonyl chloride
CAS: 2547-61-7
Molecular Formula: CCl4O2S
TRICHLOROMETHANESULFONYL CHLORIDE - Names and Identifiers
TRICHLOROMETHANESULFONYL CHLORIDE - Physico-chemical Properties
| Molecular Formula | CCl4O2S |
| Molar Mass | 217.89 |
| Density | 1.8252 (estimate) |
| Melting Point | 137-140°C(lit.) |
| Boling Point | 170°C |
| Flash Point | 63.7°C |
| Solubility | soluble in Toluene |
| Vapor Presure | 1.14mmHg at 25°C |
| Appearance | solid |
| Color | White to Almost white |
| Storage Condition | Room Temprature |
| Refractive Index | 1.536 |
| MDL | MFCD00007452 |
TRICHLOROMETHANESULFONYL CHLORIDE - Risk and Safety
| Hazard Symbols | C - Corrosive |
| Risk Codes | R34 - Causes burns R36/37 - Irritating to eyes and respiratory system. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S27 - Take off immediately all contaminated clothing. S28 - After contact with skin, wash immediately with plenty of soap-suds. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 3261 8/PG 2 |
| WGK Germany | 3 |
| Hazard Class | 8 |
| Packing Group | III |
TRICHLOROMETHANESULFONYL CHLORIDE - Reference Information
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| application | trichloromethyl sulfonyl chloride can be used as an intermediate in pharmaceutical synthesis. Perfluoroiodoane and bromoalkane and sodium dithionite undergo sulphonation deiodide and debromination under appropriate conditions, while perfluorochloroalkane cannot react under similar conditions. However, carbon tetrachloride and 1,1,1,1 trichloropolyfluoroalkane can undergo sulphonation dechlorination with sodium dithionite. Trichloromethane and trichloroiodomethane react under similar conditions to obtain sulphonated deiodization and deiodination products respectively. |
| preparation | step 1); Reaction at 25 ℃, under nitrogen atmosphere, 4.0g(26mmol)la,4.6g(26mmol) sodium thiosulfate, 2.2g sodium bicarbonate, 18mL of water and 6mL of ethylene, stirring at 25 ℃ for 6h, adding 2.3g sodium thiosulfate and 1.1g sodium bicarbonate, continuing the reaction for l0h, and evaporating the solvent under reduced pressure, the remaining solid was extracted three times with 50mL ethyl acetate, the extract was deesterified to obtain a white soft solid, and 3.9g colorless crystal 2a was obtained by isopropyl alcohol crystallization, with a yield of 73%. step 2):3.9g2a is dissolved in 15mL of water, and chlorine gas is passed at 0 ℃ for lh, white solid is precipitated, filtered, washed with water, dried, and purified by sublimation method to obtain 3.9g of white solid trichloromethyl sulfonyl chloride with 94% yield. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Trichloromethanesulfonyl Chloride Visit Supplier Webpage Request for quotationCAS: 2547-61-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Trichloromethanesulfonyl chloride Request for quotation
CAS: 2547-61-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2547-61-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: Trichloromethanesulfonyl Chloride Visit Supplier Webpage Request for quotationCAS: 2547-61-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Trichloromethanesulfonyl Chloride Visit Supplier Webpage Request for quotationCAS: 2547-61-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Trichloromethanesulfonyl chloride Request for quotation
CAS: 2547-61-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2547-61-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: Trichloromethanesulfonyl Chloride Visit Supplier Webpage Request for quotationCAS: 2547-61-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


